No products in the cart.
Hepatitis B Infection: Clinical Features and Immunological Response
What is hepatitis B and what are its complications?
Hepatitis B is a liver disease caused by the hepatitis B virus (HBV). This disease can lead to both acute and chronic infections(1,2). Acute infections usually last for a short period and may resolve on their own, but chronic infections can persist for years and potentially lead to serious complications(1,2).
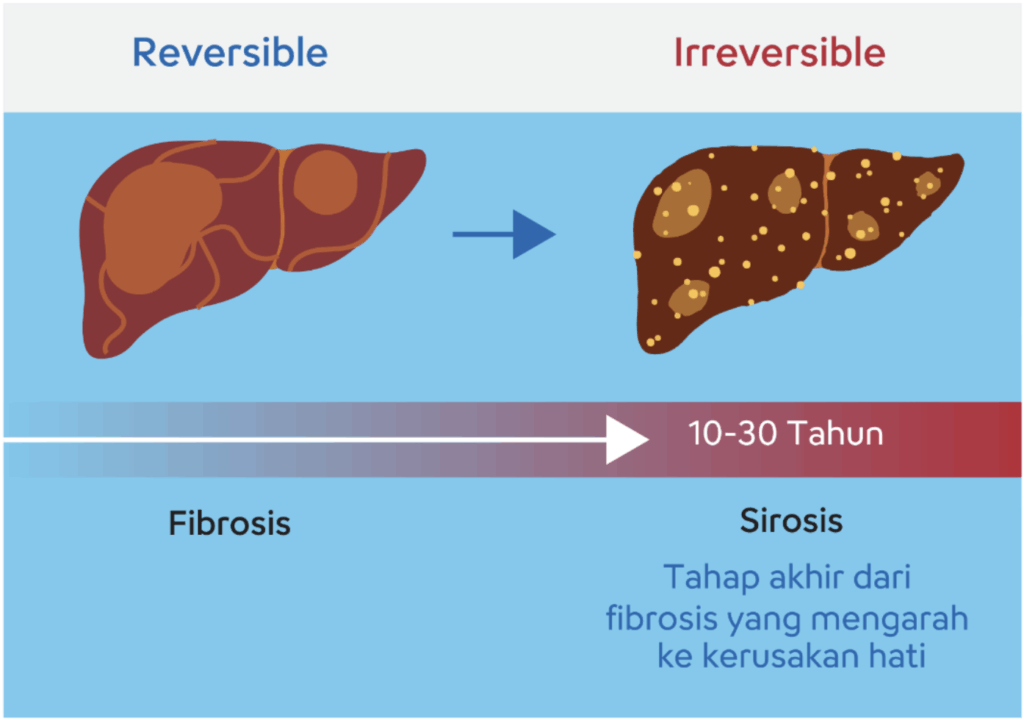
The hepatitis B virus can cause damage to liver tissue, known as fibrosis(3). Fibrosis is an early stage of liver damage where scar tissue begins to form. If left untreated, this condition can progress to cirrhosis, which is an advanced stage where the liver experiences more severe damage and liver function becomes impaired (Figure 1)(3).
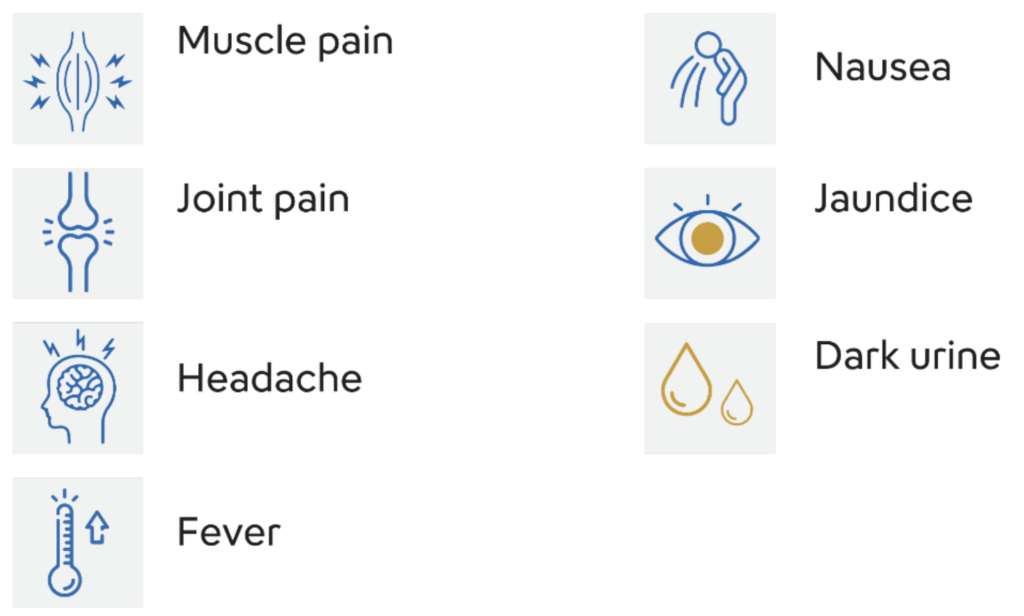
What are the symptoms and sign of hepatitis B?(2)
How is Hepatitis B diagnosed?
The diagnosis of Hepatitis B can be made using serological markers and molecular methods (Table 1). The three main serological markers used to determine HBV infection status are the hepatitis B surface antigen (HBsAg), antibodies against the hepatitis B surface antigen (anti-HBs), and antibodies against the hepatitis B core antigen (anti-HBc)(4,5,6).
In Indonesia, HBsAg testing is available at primary healthcare facilities (FKTP). Meanwhile, additional tests for Hepatitis B, such as ultrasound, liver biopsy, AFP, ALT, HBeAg, anti-HBe, and other laboratory tests, can be accessed at secondary facilities(7).

How does the immune response of the body react to Hepatitis B infection?(3)
Acute Phase
The body’s immune response to Hepatitis B infection plays a crucial role in the diagnosis and recovery from the infection. When the body is infected with HBV, the immune system begins to produce antibodies, including IgM anti-HBc, which appears in the early phase of the infection and indicates an immune response to the virus(2). In Figure 3, an acute HBV infection is depicted. During the early phase of acute HBV infection, patients show reactive results for HBsAg, IgM anti-HBc, and high levels of HBV DNA. Additionally, HBeAg may also show reactive results during this phase. As they enter the recovery phase, HBsAg becomes undetectable, and patients experience seroconversion to anti-HBs. However, there is a period of 1 to 6 months during which both HBsAg and anti-HBs are not detectable; during this phase, diagnosis relies on the presence of IgM anti-HBc. During the recovery phase, even though HBsAg becomes undetectable and anti-HBs appears, IgG anti-HBc remains present as an indicator that the body has been exposed to the virus. Therefore, serological testing that includes HBsAg, anti-HBc, and anti-HBs is not only important for diagnosis but also for understanding the body’s immune response to Hepatitis infection(2).

Fase Kronik
Pada Gambar 4 ditunjukkan infeksi HBV kronis. Dalam kondisi kronis, HBsAg terdeteksi secara persisten selama lebih dari 6 bulan, disertai dengan positifnya IgG anti-HBc. Pada fase awal infeksi, HBeAg dan tingkat DNA HBV yang tinggi dapat terdeteksi(2). Namun, pada fase-fase berikutnya, terlihat adanya anti-HBe dan variasi dalam tingkat DNA HBV. Ringkasan interpretasi penanda serologi terkait hepatitis B lebih lengkap tersedia di Tabel 22.


Steps Ahead in the Fight Against Liver Disease with Sysmex Solutions?(3)

Want to know more about Liver Disease? Click here to explore our academy
References
- US Centers for Disease Control and Prevention. (n.d.). Hepatitis B basics. https://www.cdc.gov/hepatitis-b/about/index.html
- Jeng, W.-J., Papatheodoridis, G. V., & Lok, A. S. (2023a). Hepatitis B. The Lancet, 401(10381), 1039–1052. https://doi.org/10.1016/s0140-6736(22)01468-4
- Ravndal, L., et al. (2023). Algorithms for early detection of silent liver fibrosis in the primary care setting – a scoping review. Expert Review of Gastroenterology & Hepatology, 17(10), 985–997. https://doi.org/10.1080/17474124.2023.2255522
- US Centers for Disease Control and Prevention. (n.d.). CDC Recommendation: Screening and Testing for Hepatitis B Virus Infection.https://www.cdc.gov/mmwr/volumes/ 72/rr/pdfs/rr7201a1-H.pdf
- University of Washington. (n.d.). HBV screening, testing, and diagnosis. https://www.hepatitisb.uw.edu/go/screening-diagnosis/diagnosis-hbv/core-concept/all
- Song, J. E., & Kim, D. Y. (2016). Diagnosis of hepatitis B. Annals of Translational Medicine, 4(18), 338–338. https://doi.org/10.21037/atm.2016.09.11
- Kementrian Kesehatan Republik Indonesia. (n.d.-c). Peraturan Menteri Kesehatan Republik Indonesia Nomor 53 Tahun 2015 tentang Penanggulangan Hepatitis Virus. http://hukor.kemkes.go.id/uploads/produk_hukum/PMK_No._53_ttg_Penanggulangan_Hepatitis_Virus_.pdf
- Kementrian Kesehatan Republik Indonesia. (2023). Petunjuk Teknis Manajemen Program Hepatitis B dan C. https:// cdn.who.int/ media/ docs/ default-source/ searo/ indonesia/ non-who-publications/ 2023-guideline-for-program-management-of-hepatitis-b-and-c.pdf?sfvrsn=f7373309_1&download=true
- Bui, H. H., Nguyen, S. T.-B., Phan, S. T., Nguyen, K. M., & Nguyen, C. D. (2023). Evaluating M2BPGi as a marker for liver fibrosis in patients with chronic hepatitis B. Digestive Diseases and Sciences, 68(12), 4407–4417. https://doi.org/10.1007/s10620-023-08143-5
- Tangvoraphonkchai, K., Suttichaimongkol, T., Kularbkaew, C., Sangaimwibool, P., & Sukeepaisarnjaroen, W. (2022). Application of MAC-2 binding protein glycosylation isomer as a non-invasive biomarker for probing liver disease. Scientific Reports, 12(1). https://doi.org/10.1038/s41598-022-10744-5
[/vc_column_text][/vc_column][/vc_row]
